Physical and Chemical Properties of Matter
- Page ID
- 237
We are all surrounded by matter on a daily basis. Anything that we use, touch, eat, etc. is an example of matter. Matter can be defined or described as anything that takes up space, and it is composed of miniscule particles called atoms. It must display the two properties of mass and volume.
Introduction
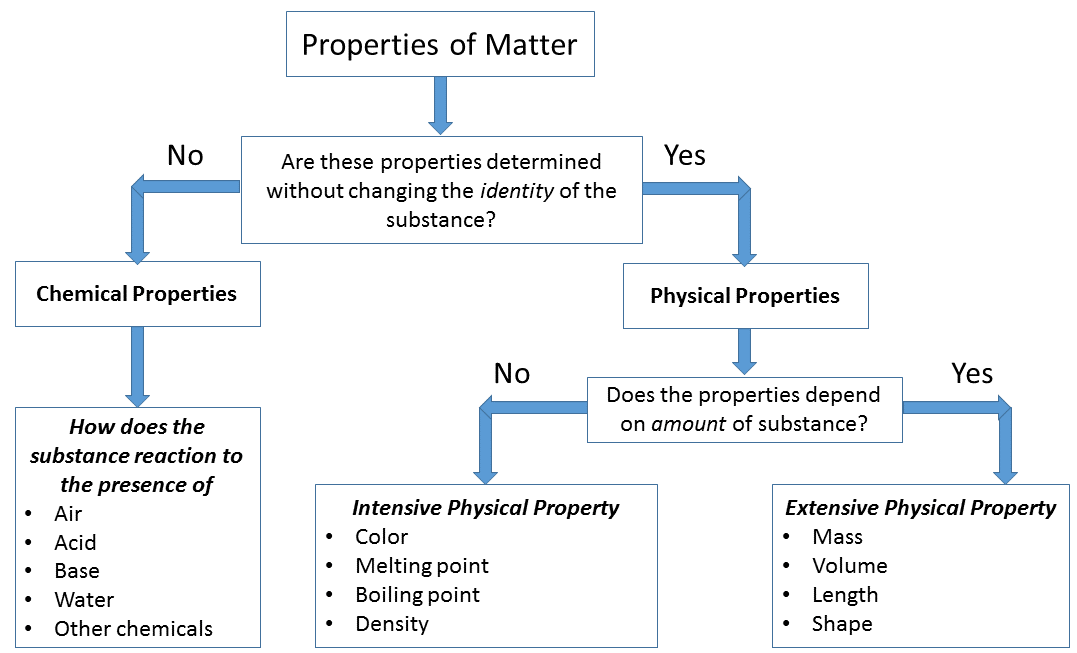
The different types of matter can be distinguished through two components: composition and properties. The composition of matter refers to the different components of matter along with their relative proportions. The properties of matter refer to the qualities/attributes that distinguish one sample of matter from another. These properties are generally grouped into two categories: physical or chemical.
Physical Properties and Changes
Physical properties can be observed or measured without changing the composition of matter. Physical properties are used to observe and describe matter. Physical properties of materials and systems are often described as intensive and extensive properties. This classification relates to the dependency of the properties upon the size or extent of the system or object in question.
An intensive property is a bulk property, meaning that it is a physical property of a system that does not depend on the system size or the amount of material in the system. Examples of intensive properties include temperature, refractive index, density, and hardness of an object. When a diamond is cut, the pieces maintain their intrinsic hardness (until their size reaches a few atoms thick). In contrast, an extensive property is additive for independent, non-interacting subsystems. The property is proportional to the amount of material in the system.
Intensive properties: A physical property that will be the same regardless of the amount of matter.
- density: \(\rho=\frac{m}{v}\)
- color: The pigment or shade
- conductivity: electricity to flow through the substance
- malleability: if a substance can be flattened
- luster: how shiny the substance looks
Extensive Properties: A physical property that will change if the amount of matter changes.
- mass: how much matter in the sample
- volume: How much space the sample takes up
- length: How long the sample is
Change in which the matter's physical appearance is altered, but composition remains unchanged.
A physical change takes place without any changes in molecular composition. The same element or compound is present before and after the change. The same molecule is present through out the changes. Physical changes are related to physical properties since some measurements require that changes be made. The three main states of matter are: Solid, Liquid, Gas
- Solid is distinguished by a fixed structure. Its shape and volume do not change. In a solid, atoms are tightly packed together in a fixed arrangement.
- Liquid is distinguished by its malleable shape (is able to form into the shape of its container), but constant volume. In a liquid, atoms are close together but not in a fixed arrangement.
- Gas is made up of atoms that are separate. However, unlike solid & liquid, a gas has no fixed shape and volume.
When liquid water (\(H_2O\)) freezes into a solid state (ice), it appears changed; However, this change is only physical as the the composition of the constituent molecules is the same: 11.19% hydrogen and 88.81% oxygen by mass.

Figure \(\PageIndex{2}\): Physical Change: Ice Melting is a physical change. from Wikipedia.
Chemical Properties and Changes
Chemical properties of matter describes its "potential" to undergo some chemical change or reaction by virtue of its composition. What elements, electrons, and bonding are present to give the potential for chemical change. It is quite difficult to define a chemical property without using the word "change". Eventually you should be able to look at the formula of a compound and state some chemical property. At this time this is very difficult to do and you are not expected to be able to do it. For example hydrogen has the potential to ignite and explode given the right conditions. This is a chemical property. Metals in general have they chemical property of reacting with an acid. Zinc reacts with hydrochloric acid to produce hydrogen gas. This is a chemical property.
Chemical change results in one or more substances of entirely different composition from the original substances. The elements and/or compounds at the start of the reaction are rearranged into new product compounds or elements. A CHEMICAL CHANGE alters the composition of the original matter. Different elements or compounds are present at the end of the chemical change. The atoms in compounds are rearranged to make new and different compounds.
Corrosion is the unwanted oxidation of metals resulting in metal oxides.
\[2 Mg + O_2 \rightarrow 2 MgO \nonumber \]

Problems
The following questions are multiple choice.
1. Milk turns sour. This is a ________________
- Chemical Change
- Physical Change
- Chemical Property
- Physical Property
- None of the above
2. HCl being a strong acid is a __________, Wood sawed in two is ___________
- Chemical Change, Physical Change
- Physical Change, Chemical Change
- Chemical Property, Physical Change
- Physical Property, Chemical Change
- None of the above
3. CuSO4 is dissolved in water
- Chemical Change
- Physical Change
- Chemical Property
- Physical Property
- None of the above
4. Aluminum Phosphate has a density of 2.566 g/cm3
- Chemical Change
- Physical Change
- Chemical Property
- Physical Property
- None of the above
5. Which of the following are examples of matter?
- A Dog
- Carbon Dioxide
- Ice Cubes
- copper (II) nitrate
- A Moving Car
6. The formation of gas bubbles is a sign of what type of change?
7. True or False: Bread rising is a physical property. 8. True or False: Dicing potatoes is a physical change. 9. Is sunlight matter? 10. The mass of lead is a _____________property.
Solutions
- chemical change
- chemical property, physical change
- physical change
- physical property
- All of the above
- chemical
- False
- True
- No
- physical property
References
- Petrucci, Bissonnette, Herring, Madura. General Chemistry: Principles and Modern Applications. Tenth ed. Upper Saddle River, NJ 07458: Pearson Education Inc., 2011.
- Cracolice, Peters. Basics of introductory Chemistry An active Learning Approach. Second ed. Belmont, CA 94001:Brooks/Cole, 2007.
Contributors and Attributions
- Samantha Ma (UC Davis)

