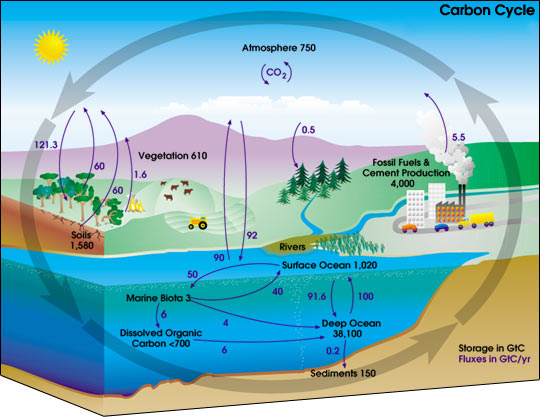
Carbon Cycle
- Page ID
- 373
Photosynthesis
Photosynthesis is a complex series of reactions carried out by algae, phytoplankton, and the leaves in plants, which utilize the energy from the sun. The simplified version of this chemical reaction is to utilize carbon dioxide molecules from the air and water molecules and the energy from the sun to produce a simple sugar such as glucose and oxygen molecules as a by product. The simple sugars are then converted into other molecules such as starch, fats, proteins, enzymes, and DNA/RNA i.e. all of the other molecules in living plants. All of the "matter/stuff" of a plant ultimately is produced as a result of this photosynthesis reaction. An important summary statement is that during photosynthesis plants use carbon dioxide and produce oxygen.
Combustion/Metabolism Reaction
Combustion occurs when any organic material is reacted (burned) in the presence of oxygen to give off the products of carbon dioxide and water and ENERGY. The organic material can be any fossil fuel such as natural gas (methane), oil, or coal. Other organic materials that combust are wood, paper, plastics, and cloth. Organic materials contain at least carbon and hydrogen and may include oxygen. If other elements are present they also ultimately combine with oxygen to form a variety of pollutant molecules such as sulfur oxides and nitrogen oxides.
Metabolism occurs in animals and humans after the ingestion of organic plant or animal foods. In the cells a series of complex reactions occurs with oxygen to convert for example glucose sugar into the products of carbon dioxide and water and ENERGY. This reaction is also carried out by bacteria in the decomposition/decay of waste materials on land and in the water.
An important summary statement is that during combustion/metabolism oxygen is used and carbon dioxide is a product. The whole purpose of both processes is to convert chemical energy into other forms of energy such as heat.
Sedimentation
Carbon dioxide is slightly soluble and is absorbed into bodies of water such as the ocean and lakes. It is not overly soluble as evidenced by what happens when a can of carbonated soda such as Coke is opened. Some of the dissolved carbon dioxide remains in the water, the warmer the water the less carbon dioxide remains in the water.
Some carbon dioxide is used by algae and phytoplankton through the process of photosynthesis.
In other marine ecosystems, some organisms such as coral and those with shells take up carbon dioxide from the water and convert it into calcium carbonate. As the shelled organisms die, bits and pieces of the shells fall to the bottom of the oceans and accumulate as sediments. The carbonate sediments are constantly being formed and redissolved in the depths of the oceans. Over long periods of time, the sediments may be raised up as dry land or into mountains. This type of sedimentary rock is called limestone. The carbonates can redissolve releasing carbon dioxide back to the air or water.
Human Impacts: Fossil Fuels
In the natural carbon cycle, there are two main processes which occur: photosynthesis and metabolism. During photosynthesis, plants use carbon dioxide and produce oxyge and during metabolism oxygen is used and carbon dioxide is a product. Humans impact the carbon cycle during the combustion of any type of fossil fuel, which may include oil, coal, or natural gas. Fossil Fuels were formed very long ago from plant or animal remains that were buried, compressed, and transformed into oil, coal, or natural gas. The carbon is said to be "fixed" in place and is essentially locked out of the natural carbon cycle. Humans intervene during by burning the fossil fuels. During combustion in the presence of air (oxygen), carbon dioxide and water molecules are released into the atmosphere. The question becomes as to what happens to this extra carbon dioxide that is released into the atmosphere. This is the subject of considerable debate and about it possible effect in enhancing the greenhouse ffect which may than result in global warming.

