Drug Receptor Interactions
- Page ID
- 415
The vast majority of drugs show a remarkably high correlation of structure and specificity to produce pharmacological effects. Experimental evidence indicates that drugs interact with receptor sites localized in macromolecules which have protein-like properties and specific three dimensional shapes. A minimum three point attachment of a drug to a receptor site is required. In most cases a rather specific chemical structure is required for the receptor site and a complementary drug structure. Slight changes in the molecular structure of the drug may drastically change specificity.
Introduction
Several chemical forces may result in a temporary binding of the drug to the receptor. Essentially any bond could be involved with the drug-receptor interaction. Covalent bonds would be very tight and practically irreversible. Since by definition the drug-receptor interaction is reversible, covalent bond formation is rather rare except in a rather toxic situation. Since many drugs contain acid or amine functional groups which are ionized at physiological pH, ionic bonds are formed by the attraction of opposite charges in the receptor site.
Polar-polar interactions as in hydrogen bonding are a further extension of the attraction of opposite charges. The drug-receptor reaction is essentially an exchange of the hydrogen bond between a drug molecule, surrounding water, and the receptor site.
Finally hydrophobic bonds are formed between non-polar hydrocarbon groups on the drug and those in the receptor site. These bonds are not very specific but the interactions do occur to exclude water molecules. Repulsive forces which decrease the stability of the drug-receptor interaction include repulsion of like charges and steric hindrance. Steric hindrance refers to certain 3-dimensional features where repulsion occurs between electron clouds, inflexible chemical bonds, or bulky alkyl groups.
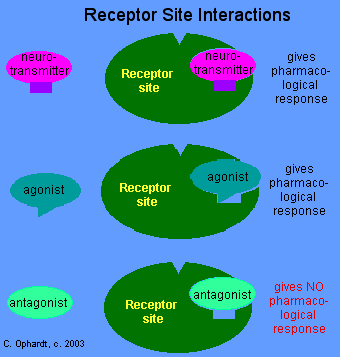
Drug Interaction with Receptor Site
- A neurotransmitter has a specific shape to fit into a receptor site and cause a pharmacological response such as a nerve impulse being sent. The neurotransmitter is similar to a substrate in an enzyme interaction. After attachment to a receptor site, a drug may either initiate a response or prevent a response from occurring. A drug must be a close "mimic" of the neurotransmitter.
- An agonist is a drug which produces a stimulation type response. The agonist is a very close mimic and "fits" with the receptor site and is thus able to initiate a response.
- An antagonist drug interacts with the receptor site and blocks or depresses the normal response for that receptor because it only partially fits the receptor site and can not produce an effect. However, it does block the site preventing any other agonist or the normal neurotransmitter from interacting with the receptor site.
Contributors
- Charles Ophardt, Professor Emeritus, Elmhurst College; Virtual Chembook

