Homework Set 1
- Page ID
- 48395
Name: ______________________________
Section: _____________________________
Student ID#:__________________________
Q1.1
Draw the molecular orbital (MO) diagrams for \(N_2\) and \(CO\). Use your MO diagrams to explain which molecule is a better \(\pi\)-acceptor when coordinated to a transition metal.
Q1.2
Draw reasonable Lewis structures (including resonance) and predict the molecular geometry for the following molecules:
- \(NO_2\)
- \(ClF_3\)
- \(OF_2\)
- \(NO\)
- \(XeO_3\)
- \(POCl_3\)
- \(XeF_2\)
- \(SF_4\)
Q1.3
Indicate if following molecules are X or L type ligands. Label each with \(\sigma\)-donor, \(\pi\)-acceptor, or \(\pi-\)donor?
- \(Ph_3P\)
- \(Cy_3P\)
- \(SCN^-\)
- \(CH_3COO^-\)
- benzyne
- \(NO\)
Q1.4
Draw and name all possible coordination modes (hapticity) of a cyclopentadienyl ligand to a transition metal.
Q1.5
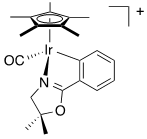
For the following complexes, provide the formal oxidation state, \(d^n\) configuration, and total valence electron count for the metal.
(a)
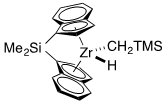
(b)
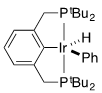
(c)
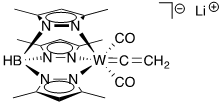
(d)
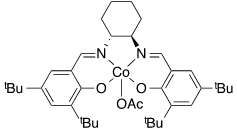
(e)
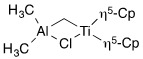
(f)
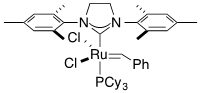
(g)
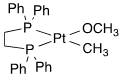
(h)