Chemistry of Cobalt
- Page ID
- 3721
Cobalt (Co) lies with the transition metals on the periodic table. The atomic number of Cobalt is 27 with an atomic mass of 58.933195. Cobalt was first discovered in 1735 by George Brandt in Stockholm Sweden. It is used in many places today, such as, magnets materials, paint pigments, glasses, and even cancer therapy. The word cobalt is from the German word kobold, which means "goblin" or "evil spirit" this term was used by miners that was really difficult to mine and harmful to the miners health.
Physical Properties
Cobalt is a sturdy, gray metal which resembles iron and nickel. Although cobalt is ductile it is also somewhat malleable. Ductile means the metals ability to be drawn into thin wires. Malleable means is the ability of being hammered into thin sheets. Next to nickel and iron, cobalt is one of the three naturally occurring magnetic metals. When cobalt is combined with another metal to make an alloy its magnetic properties are even more apparent than the individual metal of cobalt. Cobalt's melting point is 1495 degrees C with a boiling point of 2870 degrees C. The density is 8.9 grams per cubic centimeter.
| Atomic Number | 27 |
|---|---|
| Group | 9 |
| Period | 4 |
| Mass | 58.933195 g/mol |
| Density | 8.9 g.cm-3 |
| Melting Point | 1495 degrees C |
| Boiling Point | 2870 degrees C |
| Electronegativity | 1.36 |
| Specific heat | 0.568 J/mol K |
| Atomic Radius: | 1.67Å |
| Covalent Radius | 1.16Å |
| Electron Configuration | [Ar] 4s2 3d7 |
| Common Ions | 2 (most common), 3 |
| Crystal Structure | Hexagonal |
| Heat of Fusion | 16.06 kJ/mol |
| Heat of Vaporization | 377 kJ/mol |
Isotopes of Cobalt
Cobalt has one naturally occurring isotope which is cobalt 59. Isotopes are two or more forms a element. Isotopes are different because of their mass number. The number written on the right of an element i.e 59 is the mass number. The mass number is the number of protons and neutrons contained in the nucleus of the atom.The number of neutrons in a atom is how elements vary, this variation is called an isotope. There are also 10 radioactive isotopes of cobalt that are currently known. A radioactive isotope which splits apart and emits radiation. Cobalt-60 is one of the most commonly used radioactive isotopes and is used in medicine to find a treat certain diseases including the Schilling test which determines if a patients body is making and using vitamin B12 effectively. Co-57,58 are also used for the same purpose. Cobalt-60 is also used to treat cancer, because the radiation it gives off kills cancer cells.
| Co-56 | 77.3 days |
| Co-57 | 271.74 days |
| Co-58 | 70.86 days |
| Co-58m | 9.04 hours |
| Co-59 | Stable |
| Co-60 | 5.3 years |
| Co-60m | 10.467 minutes |
| Co-61 | 1.7 hours |
Applications of Cobalt
Cobalt's most important application is the production of super alloys. Super alloys consist of iron, cobalt, nickel, chromium, tungsten, aluminum, and titanium. Super alloys do not rust and retain their physical and chemical properties at high temperatures. Superalloys are used where metals are placed under a great deal of stress and high temperatures.Super alloys are commonly used in airplane parts such as jet engine components and gas turbines. Superalloys are used in gas turbines because is it is used to make electricity, which operates at a really high temperature. Cobalt is also used to make magnetic alloys. Cobalt magnetic alloys are used in devices that need to hold a magnetic field. An example of this is electric motors and generators. Cobalt can also be used for the production of cemented carbides. Cementation is the process where one metal is used to cover the coating of a second metal. Through the process of cementation a extremely hard, strong alloy such as those used in drilling tools is produced.
Compounds of Cobalt
Cobalt is relatively abundant in nature at around 10 to 30 parts per million. The common ores of cobalt include: cobaltite, smaltite, chloranthite, and linnaeite. The largest suppliers of cobalt are Zambia, Canada, Russia, Australia, Zaire, and Cuba. Cobalt it not currently mined in the United States.
Cobalt compounds are commonly used to make colored glass, glazes, paints, rubber, inks, cosmetics, and pottery. These compounds compounds include: cobalt oxide, cobalt potassium nitrite, cobalt aluminate, and cobalt ammonium phosphate. Cobalt compounds can also be used as catalyst. A catalyst is a substance that is used to speed up or lower a chemical reaction, by increasing or decreasing its activation energy. For all the cobalt used in the United States about 25% of them are cobalt compounds.
Health Effects of Cobalt
Cobalt is a trace mineral that the human body needs in only small amounts. When trace metals are absent in a diet this leads to health problems. Animals use trace minerals to make essential enzymes which function as catalysts. These enzymes speed up changes that occur in the human body. Enzymes are needed for living cells to function properly For example cobalt is needed for the natural production of B-12 vitamins. B-12 vitamins ensure that enough red blood cells are produced in the human body. Cobalt also affects other animals because of its lack of presence in the soil. For example sheep in Australia become infected with a disease called Coast disease, caused by cobalt deficiency. Excess cobalt can also lead to health problems. People who work around cobalt can inhale its dust which causes vomiting, diarrhea, or breathing problems. If cobalt is presence on the skin in can lead to an irritation and rash.
Chemical Properties
Cobalt is somewhat of a reactive element. It combines with oxygen in the air, but does not catch on fire and burn unless it is in powder form. Cobalt has the ability to react with most acids to produce hydrogen gas. However cobalt does not react with water that is at room temperature. The simplest ion that cobalt forms in solution is the pink hexaaquacobalt(II) ion - [Co(H2O)6]2+. Cobalt(II) chloride is often used in humidity indicators because in dry form it is blue and when hydrated it is pink. The radioactive isotope of cobalt, Co-60, is used in portable "x-ray" devices. It actually produces gamma rays which can be used to detect internal flaws in metal in much the same way as x-rays by producing a shadowy image on film. Co-60 is also used in cancer treatment.
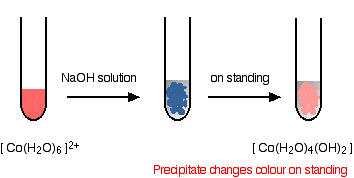
Ligand Exchange of hexaaquacobalt(II) ions with hydroxide ions
Hydroxide ions remove hydrogen ions from the water ligands attached to the cobalt ion. Once a hydrogen ion has been removed from two of the water molecules, you are left with a complex with no charge - a neutral complex. This is insoluble in water and a precipitate is formed.
\[ \ce{ [Co(H2O)6]^{2+} + 2OH^{-} \rightarrow [Co(H2O)4(OH)2] + 2H2O} \nonumber \]
In the test-tube, the color changes are:
Ligand Exchange of hexaaquacobalt(II) ions with ammonia solution
The ammonia acts as both a base and a ligand. With a small amount of ammonia, hydrogen ions are pulled off the hexaaqua ion exactly as in the hydroxide ion case to give the same neutral complex.
\[ [Co(H_2O)_6]^{2+} + 2NH_3 \rightarrow [Co(H_2O)_4(OH)_2] + 2NH_4^+ \nonumber \]
That precipitate dissolves if you add an excess of ammonia. The ammonia replaces water as a ligand to give hexaamminecobalt(II) ions.
\[ [Co(H_2O)_6]^{2+} + 6NH_3 \rightarrow [Co(NH_3)_6]^{2+} + 6H_2O \nonumber \]
The color changes are:
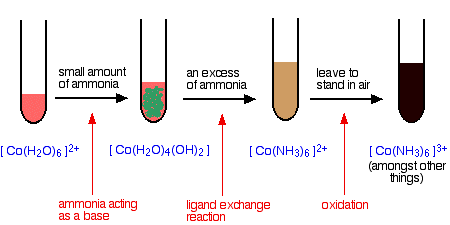
The hexaamminecobalt(II) complex is very easily oxidized to the corresponding cobalt(III) complex. In the test-tube this is seen as a rapid darkening to a deep red-brown solution. In fact the hexaamminecobalt(III) ion is yellow! What you see is a mixture of this ion and various other cobalt(III) ions involving ligand exchange reactions with both water molecules and negative ions present in the solution.
Another oxidation of hexaaquacobalt(II) ions
Rather than relying on oxidation by the air, you can add an oxidizing agent such as hydrogen peroxide. You can do this after the addition of ammonia as in the last case, or you can do it following addition of sodium hydroxide solution.
With ammonia solution and hydrogen peroxide
The reaction with ammonia solution followed by hydrogen peroxide produces the same dark reddish-brown solution as before - only faster. The equation for the oxidation of the a hexammine complex is:
\[ 2Co(NH_3)_6^{2+} + H_2O_2 \rightarrow 2Co(NH_3)_6^{3+} + 2OH^- \nonumber \]
With sodium hydroxide solution and hydrogen peroxide
You get the variably colored precipitate of the cobalt(II) hydroxide complex when you add the sodium hydroxide solution. Addition of hydrogen peroxide produces lots of bubbles of oxygen and a dark chocolate brown precipitate.
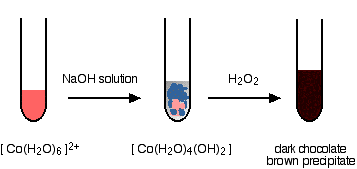
The final precipitate contains cobalt in the +3 oxidation state.
The nature of the precipate of thie reaction is under. One resource describes it as a "hydrous \(Co_2O_3\), i.e., cobalt(III) oxide with closely associated water. Another suggests a the priciptates is \(CoO(OH)\). The oxygen seen in the reaction is produced from the decomposition of the hydrogen peroxide in a side reaction. Many things catalyze this decomposition - presumably, in this case, one or more of the various cobalt compounds present.
The reaction of hexaaquacobalt(II) ions with carbonate ions
You simply get a precipitate of what you can think of as cobalt(II) carbonate.
\[ Co^{2+} (aq) + CO_3^{2-} (aq) \rightarrow CoCO_3 (s) \nonumber \]
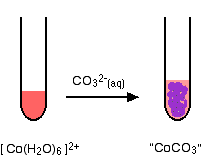
The the precipitate is better described as a basic carbonate with a formula of the type \(xCoCO_3,yCo(OH)_2,zH_2O\). A greater discussion of general reactions between hexaaqua ions and carbonate ions is discussed elsewhere.
A ligand exchange reaction involving chloride ions
If you add concentrated hydrochloric acid to a solution containing hexaaquacobalt(II) ions, the solution turns from its original pink color to a rich blue. The six water molecules are replaced by four chloride ions.
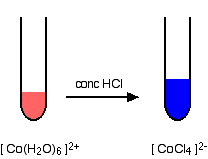
The reaction taking place is reversible.
\[ [Co(H_2O)_6]^{2+} + 4Cl^- \rightleftharpoons [CoCl_4]^{2-} + 6H_2O \nonumber \]
If you add water to the blue solution, it returns to the pink color. This reaction is explored in more detail on the module addressing ligand exchange reactions.
References
- Petrucci et al. General Chemistry: Principles & Modern Applications, 9th Edition. New Jersey: Pearson Education, Inc., 2007.
- Newton, David. Chemical Elements. 2008-04, 2008. Print
- "Highly Active Iron and Cobalt Catalysts for the Polymerization of Ethylene - Journal of the American Chemical Society (ACS Publications)." ACS Publications - Cookie Absent. Web. 02 June 2010. <http://pubs.acs.org/doi/abs/10.1021/ja9802100>.
- "Cobalt-catalysed Growth of Carbon Nanotubes with Single-atomic-layer Walls." Nature Publishing Group : Science Journals, Jobs, and Information. Web. 02 June 2010. <www.nature.com/nature/journal.../363605a0.html>
- "Giant Magnetic Anisotropy of Single Cobalt Atoms and Nanoparticles -- Gambardella Et Al. 300 (5622): 1130 -- Science." Science/AAAS | Scientific Research, News and Career Information. Web. 02 June 2010. <www.sciencemag.org/cgi/conten.../300/5622/1130>.
- "Highly Active Iron and Cobalt Catalysts for the Polymerization of Ethylene - Journal of the American Chemical Society (ACS Publications)." ACS Publications - Cookie Absent. Web. 02 June 2010. <http://pubs.acs.org/doi/abs/10.1021/ja9802100>.
- "Magnetism from the Atom to the Bulk in Iron, Cobalt, and Nickel Clusters -- Billas Et Al. 265 (5179): 1682 -- Science." Science/AAAS | Scientific Research, News and Career Information. Web. 02 June 2010. <www.sciencemag.org/cgi/conten.../265/5179/1682>.
- In Scopus. "ScienceDirect - Journal of Catalysis : Formation of Filamentous Carbon from Iron, Cobalt and Chromium Catalyzed Decomposition of Acetylene." ScienceDirect - Home. Web. 02 June 2010. www.sciencedirect.com/science...fbd00b5adf786f
Problems
- Is Cobalt ferromagnetic or paramagnetic?
- What is the electron configuration of Co+?
- Who invented Cobalt? Where?
- Why is Cobalt-60 used to treat cancer?
- What are cobalt compounds used to make?
- Cobalt reacts with what to produce hydrogen gas?
- When is superalloys use preferred?
- What are the common ores of cobalt?
- Name one of the uses of cobalt-60, other than cancer treatment.
- Describe the derivation of the name cobalt.
Solutions
- Cobalt is ferromagnetic
- [Ar]4s23d6
- George Brandt in Stockholm Sweden
- Cobalt-60 is used to treat cancer because the radiation it gives off kills cancer cells.
- Cobalt compounds are commonly used to make colored glass, glazes, paints, rubber, inks, cosmetics, and pottery.
- Cobalt reacts with most acids to produce hydrogen gas.
- Superalloys are used where metals are placed under a great deal of stress and high temperatures.
- The common ores of cobalt include: cobaltite, smaltite, chloranthite, and linnaeite
- Cobalt-60 is used in the Schilling test which determines if a patients body is making and using vitamin B12 effectively.
- The word cobalt is from the German word Kobold, which means "goblin" or "evil spirit" this term was used by miners that was really difficult to mine and harmful to the miners health
Contributors and Attributions
Jim Clark (Chemguide.co.uk)
- Christine Han (UC Davis), Harjeet Bassi (UCD)


