Nomenclature of Alcohols
- Page ID
- 1164
In the IUPAC system of nomenclature, functional groups are normally designated in one of two ways. The presence of the function may be indicated by a characteristic suffix and a location number. This is common for the carbon-carbon double and triple bonds which have the respective suffixes -ene and -yne. Halogens, on the other hand, do not have a suffix and are named as substituents, for example: (CH3)2C=CHCHClCH3 is 4-chloro-2-methyl-2-pentene.
Alcohols are usually named by the first procedure and are designated by an -ol suffix, as in ethanol, CH3CH2OH (note that a locator number is unnecessary on a two-carbon chain). On longer chains the location of the hydroxyl group determines chain numbering. For example: (CH3)2C=CHCH(OH)CH3 is 4-methyl-3-penten-2-ol. Other examples of IUPAC nomenclature are shown below, together with the common names often used for some of the simpler compounds. For the mono-functional alcohols, this common system consists of naming the alkyl group followed by the word alcohol. Alcohols may also be classified as primary, 1º, secondary, 2º, and tertiary, 3º, in the same manner as alkyl halides. This terminology refers to alkyl substitution of the carbon atom bearing the hydroxyl group (colored blue in the illustration).
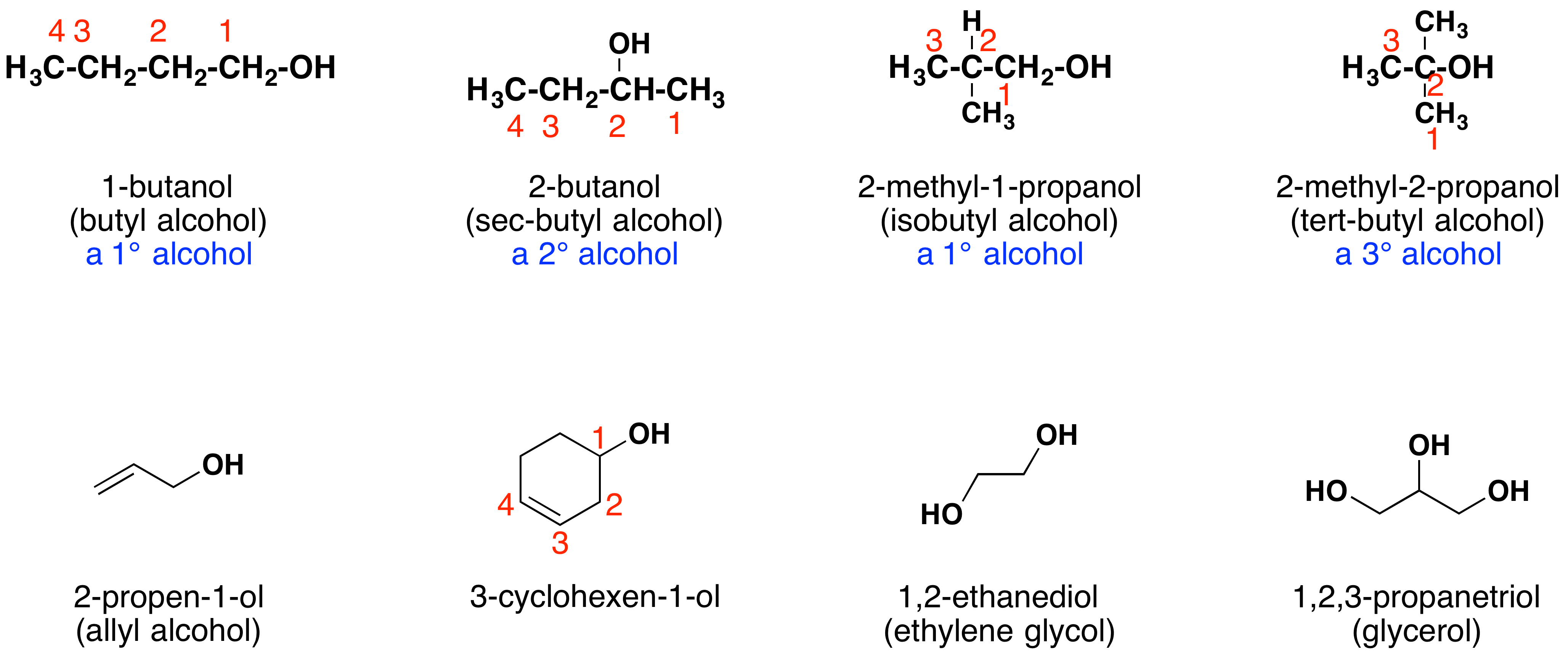
Many functional groups have a characteristic suffix designator, and only one such suffix (other than "-ene" and "-yne") may be used in a name. When the hydroxyl functional group is present together with a function of higher nomenclature priority, it must be cited and located by the prefix hydroxy and an appropriate number. For example, lactic acid has the IUPAC name 2-hydroxypropanoic acid.
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry


