Haloalkanes
- Page ID
- 856
The haloalkanes, also known as alkyl halides, are a group of chemical compounds comprised of an alkane with one or more hydrogens replaced by a halogen atom (fluorine, chlorine, bromine, or iodine).
There is a fairly large distinction between the structural and physical properties of haloalkanes and the structural and physical properties of alkanes. As mentioned above, the structural differences are due to the replacement of one or more hydrogens with a halogen atom. The differences in physical properties are a result of factors such as electronegativity, bond length, bond strength, and molecular size.
Halogens and the Character of the Carbon-Halogen Bond
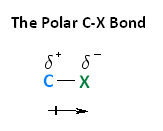
With respect to electronegativity, halogens are more electronegative than carbons. This results in a carbon-halogen bond that is polarized. As shown in the image below, carbon atom has a partial positive charge, while the halogen has a partial negative charge.
The following image shows the relationship between the halogens and electronegativity. Notice, as we move up the periodic table from iodine to fluorine, electronegativity increases.
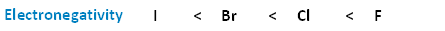
The following image shows the relationships between bond length, bond strength, and molecular size. As we progress down the periodic table from fluorine to iodine, molecular size increases. As a result, we also see an increase in bond length. Conversely, as molecular size increases and we get longer bonds, the strength of those bonds decreases.

Haloalkanes Have Higher Boiling Points than Alkanes
When comparing alkanes and haloalkanes, we will see that haloalkanes have higher boiling points than alkanes containing the same number of carbons. London dispersion forces are the first of two types of forces that contribute to this physical property. You might recall from general chemistry that London dispersion forces increase with molecular surface area. In comparing haloalkanes with alkanes, haloalkanes exhibit an increase in surface area due to the substitution of a halogen for hydrogen. The incease in surface area leads to an increase in London dispersion forces, which then results in a higher boiling point.
Dipole-dipole interaction is the second type of force that contributes to a higher boiling point. As you may recall, this type of interaction is a coulombic attraction between the partial positive and partial negative charges that exist between carbon-halogen bonds on separate haloalkane molecules. Similar to London dispersion forces, dipole-dipole interactions establish a higher boiling point for haloalkanes in comparison to alkanes with the same number of carbons.

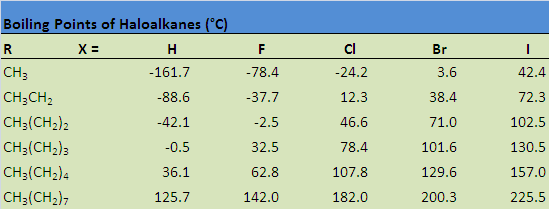
The table below illustrates how boiling points are affected by some of these properties. Notice that the boiling point increases when hydrogen is replaced by a halogen, a consequence of the increase in molecular size, as well as an increase in both London dispersion forces and dipole-dipole attractions. The boiling point also increases as a result of increasing the size of the halogen, as well as increasing the size of the carbon chain.
Contributors
- Rachael Curtis (UC Davis)

