II. Atom Abstraction
- Page ID
- 23820
A. Halogen-Atom Abstraction
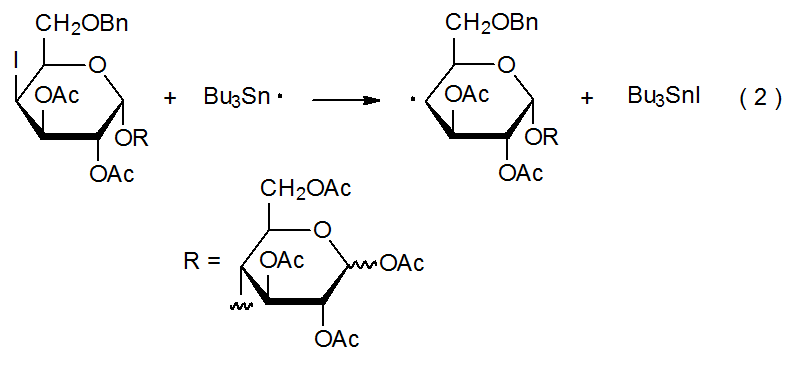
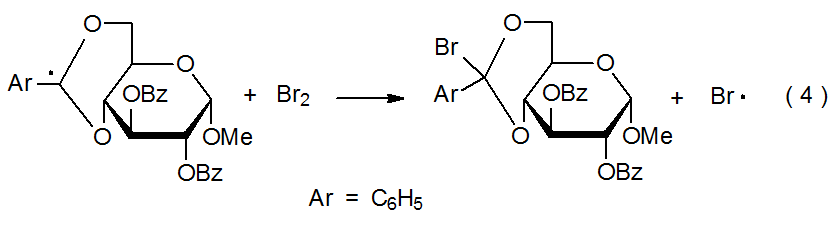
Halogen-atom abstraction can take place both in forming a halogenated carbohydrate and in removing a halogen atom from such a compound. When abstraction is from a halogenated carbohydrate, it produces a carbohydrate radical (eq 1). The abstracting radical typically is tin-centered or silicon-centered. In the reaction shown in eq 2, for example, a carbohydrate radical forms when a tin-centered abstracts an iodine atom from a deoxyiodo sugar.3 Abstraction that generates a halogenated carbohydrate takes place when I2, Br2, or another halogen donor reacts with a carbohydrate radical (eq 3). Equation 4 describes a reaction of this type.4
.png?revision=1&size=bestfit&width=295&height=115)
.png?revision=1&size=bestfit&width=395&height=184)
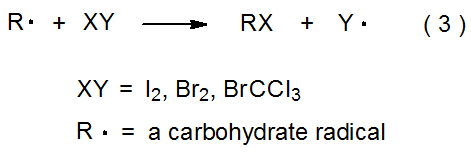.png?revision=1&size=bestfit&width=235&height=79)
.png?revision=1&size=bestfit&width=420&height=115)
B. Hydrogen-Atom Abstraction
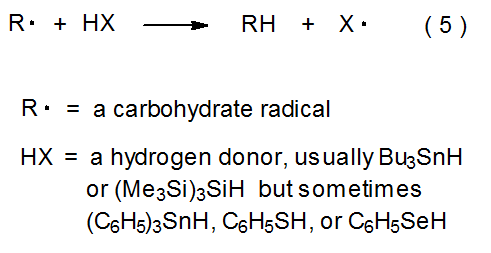
Hydrogen-atom abstraction is an elementary reaction that permeates the free-radical chemistry of carbohydrates. Because it is the final propagation step in many chain reactions, hydrogen-atom abstraction often converts a carbon-centered radical into a stable product. The hydrogen-atom donor in such reactions usually is a tin or silicon hydride, but sometimes a thiol or selenol serves in this role (eq 5). The final step in the simple reduction shown in eq 6 is a typical, hydrogen-atom-abstraction reaction.3
.png?revision=1&size=bestfit&width=245&height=126)
.png?revision=1&size=bestfit&width=325&height=197)
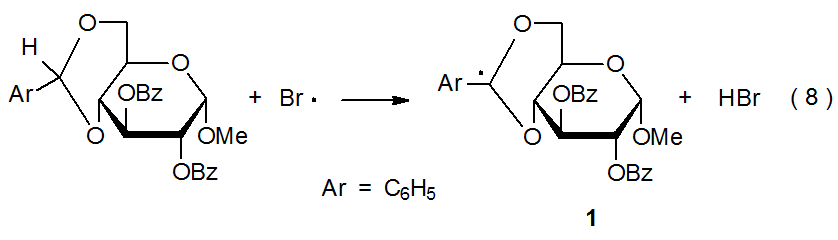
Carbohydrates also can serve as hydrogen atom donors (eq 7). A radical centered on a bromine, chlorine, or oxygen atom (and, sometimes, on a sulfur or carbon atom) is able to abstract a hydrogen atom from a carbohydrate in an elementary reaction that can be highly regioselective. For intermolecular reactions this selectivity is due to radicals preferentially abstracting the hydrogen atoms that produce the most stable carbon-centered radicals. In the reaction shown in eq 8, for example, the bromine atom abstracts only the hydrogen atom that produces the highly resonance-stabilized radical 1.4
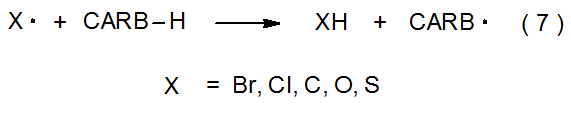.png?revision=1&size=bestfit&width=285&height=57)
.png?revision=1&size=bestfit&width=420&height=120)
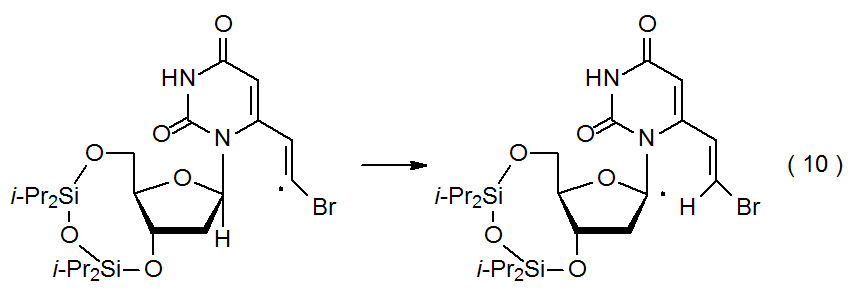
If a radical is centered on an oxygen or carbon atom in a carbohydrate, internal abstraction becomes a possibility. Such abstraction is regioselective not only because a more stable radical is being produced but also because the radical center is able easily to come within bonding distance of a limited number of hydrogen atoms (sometimes only one). In the reaction shown in eq 9, the only hydrogen atom abstracted is the one that is 1,6-related to the radical center.5 Although an oxygen-centered radical (e.g., 2 in eq 9) is reactive enough to abstract a hydrogen atom from any carbon-hydrogen bond in a carbohydrate,6 only the most reactive carbon-centered radicals (e.g., primary and vinylic ones) are capable of such reaction (eq 10).7
.png?revision=1&size=bestfit&width=240&height=125)
.png?revision=1&size=bestfit&width=425&height=150)

