VIII. Disproportionation
- Page ID
- 23824
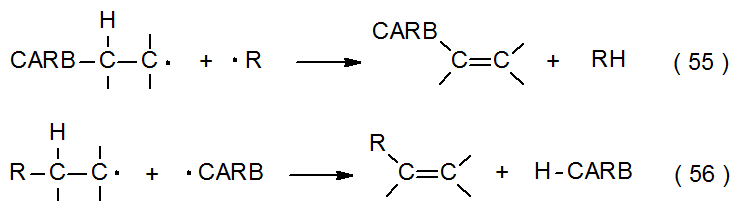
Disproportionation involving carbohydrate radicals (eq 55 and eq 56) is similar to radical combination in that most such radicals do not exist long enough in solution to come into contact with each other before another reaction takes place. As mentioned in the previous section, one situation in which two radicals can interact is when locally high concentrations are created by electrolysis; thus, when the radicals shown in eq 57 come within bonding distance, both disproportionation (10%-20%) and radical combination (69%) take place.42 In contrast, disproportionation is the exclusive process in the reaction shown in eq 58 because there is considerable thermodynamic gain from forming a highly resonance-stabilized ketone while avoiding the hindrance inherent in the combination of sterically demanding radicals.43
%252C_(56).png?revision=1&size=bestfit&width=370&height=107)
.png?revision=1&size=bestfit&width=440&height=200)
.png?revision=1&size=bestfit&width=420&height=106)

