VII. Silyl Ether Rearrangement
- Page ID
- 24027
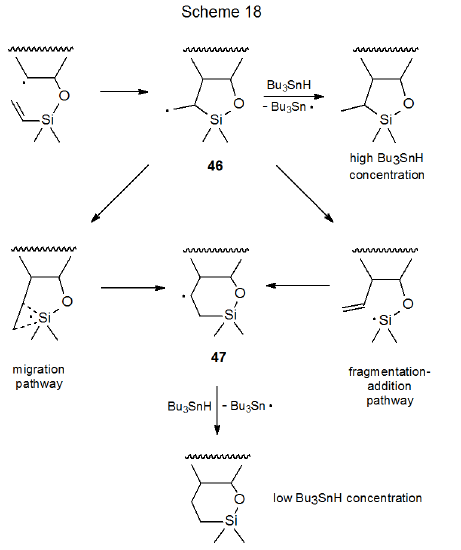
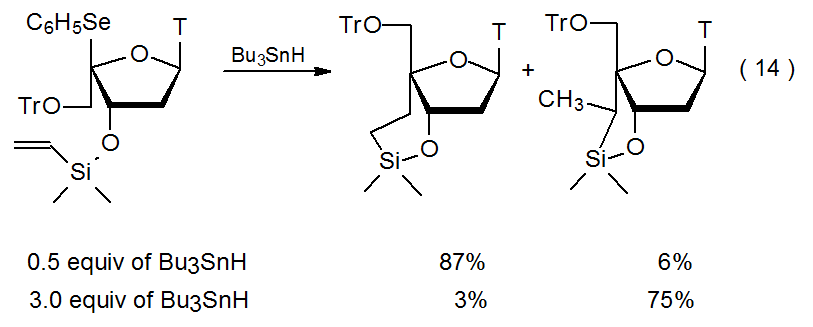
Rearrangement takes place during radical cyclization involving some silyl ethers. The primary evidence for this rearrangement is the dependence of product ring size on the concentration of Bu3SnH, the hydrogen-atom transfer in these reactions. When the reaction shown in eq 14 is conducted in dilute Bu3SnH solution, the major product contains a six-membered ring,35 but at high Bu3SnH concentration reaction regioselectivity changes to give a product with a five‑membered ring.36,37 This concentration dependence can be explained by the more rapidly formed, but less stable, radical 46 having sufficient time and energy, when the concentration of Bu3SnH is low, to be converted into the more stable radical 47, either by a rearrangement that involves a cyclic transition state or by a fragmentation-addition sequence (Scheme 18).37 At high Bu3SnH concentration hydrogen-atom abstraction occurs before ring expansion can take place.
.png?revision=1&size=bestfit&width=410&height=165)