Ether Synthesis
- Page ID
- 831
Ethers are usually prepared from alcohols or their conjugate bases. One important procedure, known as the Williamson Ether Synthesis, proceeds by an SN2 reaction of an alkoxide nucleophile with an alkyl halide. Reactions #1 and #2 below are two examples of this procedure. When applied to an unsymmetrical ether, as in this case, there are two different combinations of reactants are possible. Of these one is usually better than the other. Since alkoxide anions are strong bases, the possibility of a competing E2 elimination must always be considered. Bearing in mind the factors that favor substitution over elimination, a 1º-alkyl halide should be selected as a preferred reactant whenever possible. Thus, reaction #1 gives a better and cleaner yield of benzyl isopropyl ether than does reaction #2, which generates considerable elimination product.
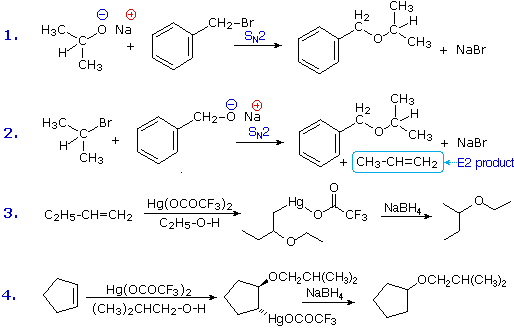
A second general ether synthesis, alkoxymercuration, is patterned after the oxymercuration reaction. Reactions #3 and #4 are examples of this two-step procedure. Note that the alcohol reactant is used as the solvent, and a trifluoroacetate mercury (II) salt is used in preference to the acetate (trifluoroacetate anion is a poorer nucleophile than acetate). The mechanism of alkoxymercuration is similar to that of oxymercuration, with an initial anti-addition of the mercuric species and alcohol being followed by reductive demercuration.
Acid-catalyzed dehydration of small 1º-alcohols constitutes a specialized method of preparing symmetrical ethers. As shown in the following two equations, the success of this procedure depends on the temperature. At 110º to 130 ºC an SN2 reaction of the alcohol conjugate acid leads to an ether product. At higher temperatures (over 150 ºC) an E2 elimination takes place.
| 2 CH3CH2-OH + H2SO4 | 130 ºC |
CH3CH2-O-CH2CH3 + H2O |
| CH3CH2-OH + H2SO4 | 150 ºC |
CH2=CH2 + H2O |
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry


