Resonance Forms
- Page ID
- 842
A resonance form is another way of drawing a Lewis dot structure for a given compound. Equivalent Lewis structures are called resonance forms. They are used when there is more than one way to place double bonds and lone pairs on atoms. Resonance structures arise when there are more than one way to draw a Lewis dot diagram that satisfies the octet rule. Remember the octet rule is where the atom gains, loses, or shares electrons so that the outer electron shell has eight electrons. We draw them when one structure does not accurately show the real structure.
Introduction
There are some basic principle on the resonance theory. First resonance structures are not real, they just show possible structures for a compound. Resonance structures are not in equilibrium with each other. Resonance structures are not isomers. Isomers have different arrangement of both atoms and electrons. Resonance forms differ only in arrangement of electrons.
Resonance structures are a better depiction of a Lewis dot structure because they clearly show bonding in molecules. Not all resonance structures are equal there are some that are better than others. The better ones have minimal formal charges, negative formal charges are the most electronegative atoms, and bond is maximized in the structure. The more resonance forms a molecule has makes the molecule more stable. They are drawn with a double-headed arrow between them to show the actual structure is somewhere between the resonance structures. These structures used curved arrow notation to show the movement of the electrons in one resonance form to the next.
Formal charges are used in Chemistry to determine the location of a charge in a molecule and determine how good of a Lewis structure it will be. Remember, the best resonance structure is the one with the least formal charge. This is why formal charges are very important. Atoms that are missing one or more electrons will have a positive charge. An atom with many electrons will have a negative charge. Assigning formal charges to an atom is very useful in resonance forms.
Formal charge is calculated using this format:
# of valence electrons- (#non bonding electrons + 1/2 #bonding electrons)
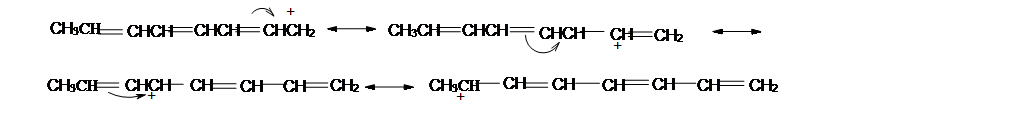
Curved arrow notation is used in showing the placement of electrons between atoms. The tail of the arrow begins at the electron source and the head points to where the electron will be. Make sure the arrows are clear including the single and half headed arrow. The reader must know the flow of the electrons.
Common Examples
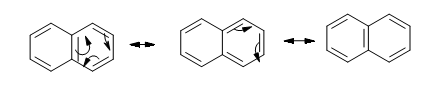
Benzene is commonly seen in Organic Chemistry and it has a resonance form. Benzene has two resonance structures, showing the placements of the bonds.

Another example of resonance is ozone. Ozone is represented by two different Lewis structures. The difference between the two structures is the location of double bond.
Drawing Resonance Forms
There are several things that should be checked before and after drawing the resonance forms. First know where the nonbonding electrons are, keep track of formal charges on atoms, and do not break sigma bonds. Finally, after drawing the resonance form make sure all the atoms have eight electrons in the outer shell. Checking these will make drawing resonance forms easier.
When drawing a resonance structure there are three rules that need to be followed for the structures to be correct:
- Only electrons move and the nuclei of the atoms never move.
- Only electrons that can move are pi electrons, single unpaired electrons, and lone pair electrons.
- The total number of electrons in the molecule do not change and neither do the number of paired and unpaired electrons.
Approaches for moving electrons are move pi electrons toward a positive charge or toward an another pi bond. Move a single nonbonding electron towards a pi bond. Move lone pair electrons toward a pi bond and when electrons can be moved in more than one direction, move them to the more electronegative atom.
Helpful hints
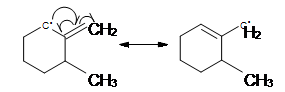
- Two resonance structures differ in the position of multiple bonds and non bonding electron. The placement of atoms and single bonds always stays the same.
- They must make sense and agree to the rules. Hydrogens must have two electrons and elements in the second row cannot have more than 8 electrons. If so, the resonance structure is not valid. Always look at the placement of arrows to make sure they agree.
- Electrons move toward a sp2 hybridized atom. The sp2 hybridized atom is either a double-bonded carbon, or a carbon with a positive charge, or it is an unpaired electron. Electrons do not move toward a sp3 hybridized carbon because there is no room for the electrons.
After drawing resonance structures check the net charge of all the structures. For example, if a structure has a net charge of +1 then all other structures must also have a net charge of +1. If not, the structure is not correct. Always check the net charge after each structure. These important details can ensure success in drawing any Resonance structure.
Outside Links
References
- McMurry, John M. Organic Chemisry A Biological Approach. N.p.: Thomson, 2007.