16.7: NAD(P)H, FADH2 and metabolism - a second look
- Page ID
- 1015
After concentrating for a good part of this chapter on the mechanistic details of how the nicotinamide and flavin coenzymes participate in the oxidation and reduction of biological organic molecules, it is worthwhile at this point to take a step back and look again at the bigger picture of how these reactions fit into the overall metabolic systems of living things. We know that cells obtain energy by breaking down and oxidizing carbohydrates, fats and proteins, ultimately transferring electrons from these fuel molecules to molecular oxygen to form water and carbon dioxide. In the opposite direction, we also know that cells need a source of electrons in order to carry out the synthesis of large, reduced molecules. How do the reactions we have been studying fit into this bigger picture?
16.7A: NADH and FADH2 as carriers of hydrides from fuel molecules to water
First, let's consider the oxidation of fuel molecules, also known as catabolism. In these processes, you can think of NADH and FADH2 as shuttles, carrying electrons - in the form of hydride ions -from fuel molecules to oxygen. Here are just a few catabolic reactions in which NADH and FADH2 are formed from NAD+ and FAD, respectively:

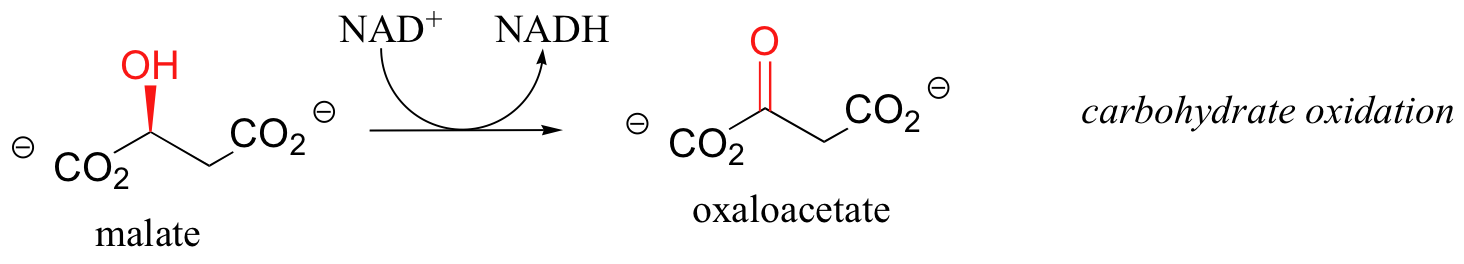
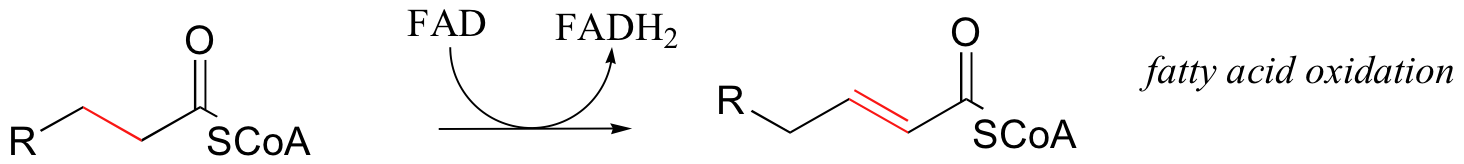

The common theme in all of these reactions is that in each case, NAD+ and FAD have accepted H2 - two high-energy electrons and two protons - from an organic substrate that is being oxidized. NADH and FADH2 then serve as conduits in the transfer of these electrons to molecular oxygen, reducing it to water.

The actual process by which electrons flow from NADH and FADH2 to water, and also by which the energy thereby released is converted into ATP, is a long, complicated multi-enzyme pathway called the 'electron-transport chain' of 'oxidative phosphorylation'. You will learn much more about this process if you take a course in biochemistry, and, because metal-binding enzymes play a central role in many of the reactions, you may also study the electron-transport enzymes in a more advanced bioinorganic chemistry course. But for now, you have a good understanding of the organic chemistry involved in the very beginning part of the process, in which electrons start their journey by being transferred as hydrides from fuel molecules to NAD+ or FAD.
16.7B: The source of NADPH for reductive biosynthesis
What about the biosynthetic, or anabolic, side of the equation? The cells of all organisms require NADPH, the primary biological reducing agent, to build large reduced molecules from small, oxidized precursors. In almost all known forms of life (we will discuss the exception shortly), NADPH is generated by two relatively simple reactions in what is known as the 'oxidative pentose phosphate pathway'. Here, the hemiacetal group of glucose-6-phosphate is first oxidized to form a lactone (a cyclic ester). This oxidation (step 1)) generates one molecule of NADPH that can be used in reductive biosynthetic reactions.
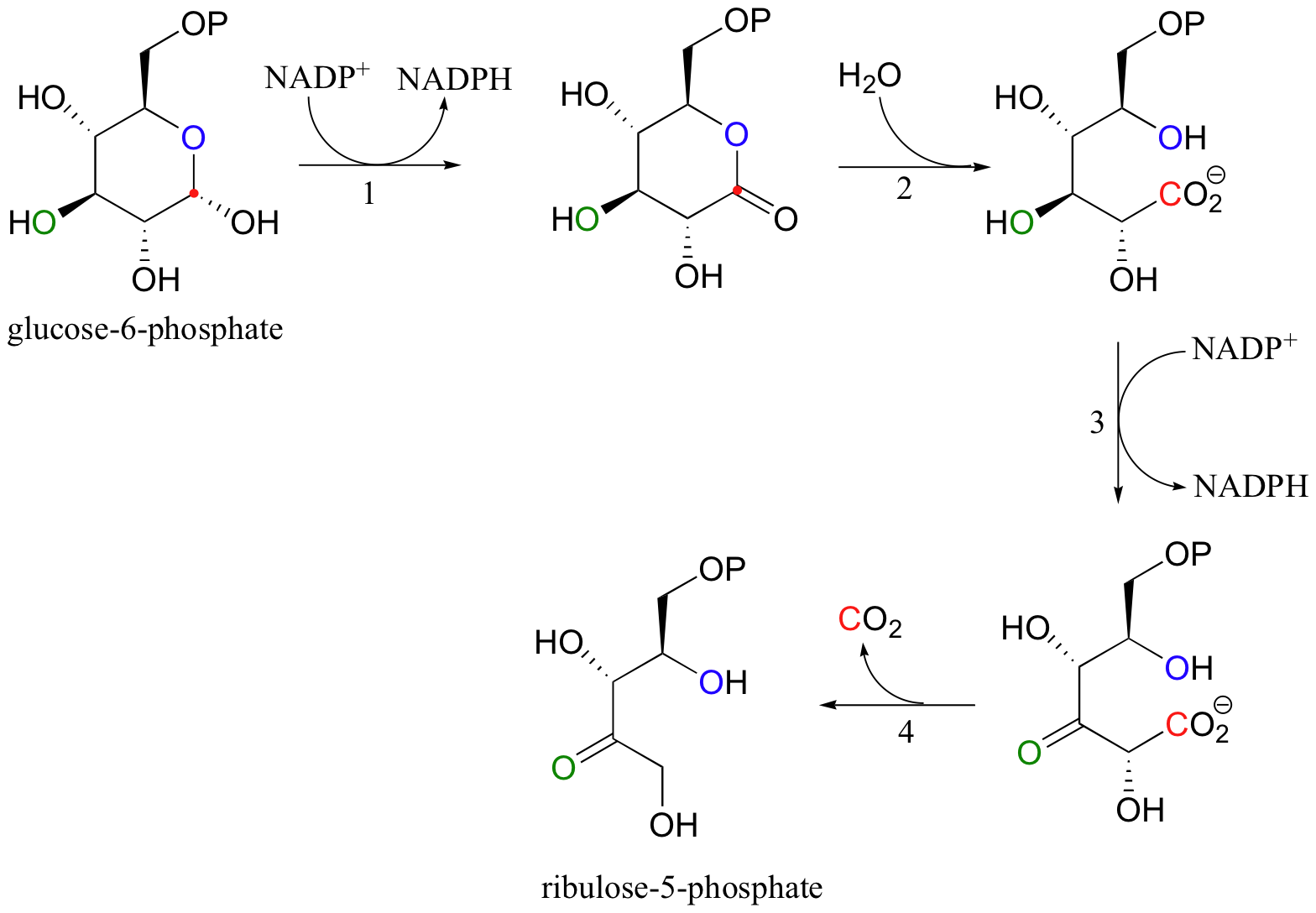
The lactone group is then hydrolized (step 2), and a second molecule of NADPH is generated in a alcohol to ketone oxidative reaction (step 3), which is quickly followed by a decarboxylation (step 4). The remaining five-carbon sugar is ribulose-5-phosphate, which, after stereoisomerization to ribose-5-phosphate (section 13.2B), serves as the foundation for the construction of DNA and RNA nucleotides.
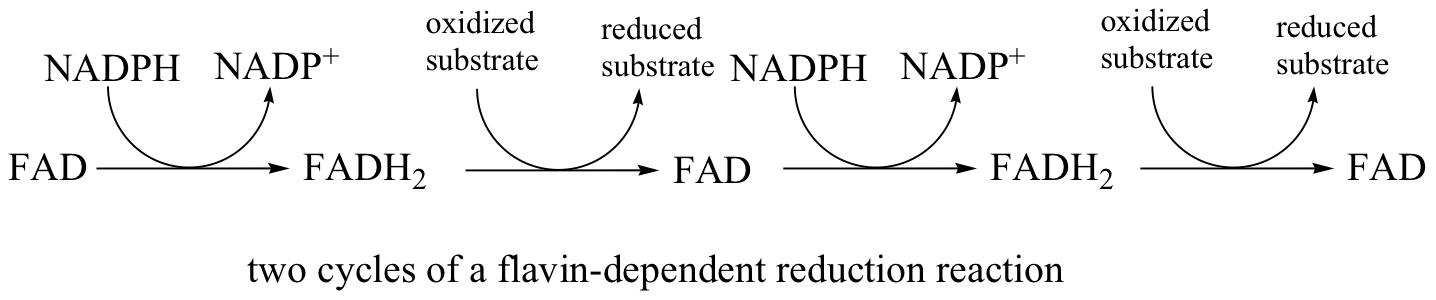
In addition to NADPH, flavin coenzyme in the form of FADH2 (and sometimes FMNH2) can serve as the immediate reducing agent in biochemical reactions. We have already seen an example of FMNH2 acting as the reducing agent in an alkene hydrogenation reaction (section 16.5C), and later in this chapter we will discuss reactions where FADH2 participates directly in the reduction disulfides to thiols (section 16.12). While the flavin coenzyme is the direct source of hydrides in these reductive reactions, the hydrides originally come from NADPH, which was formed in the oxidative pentose phosphate pathway.
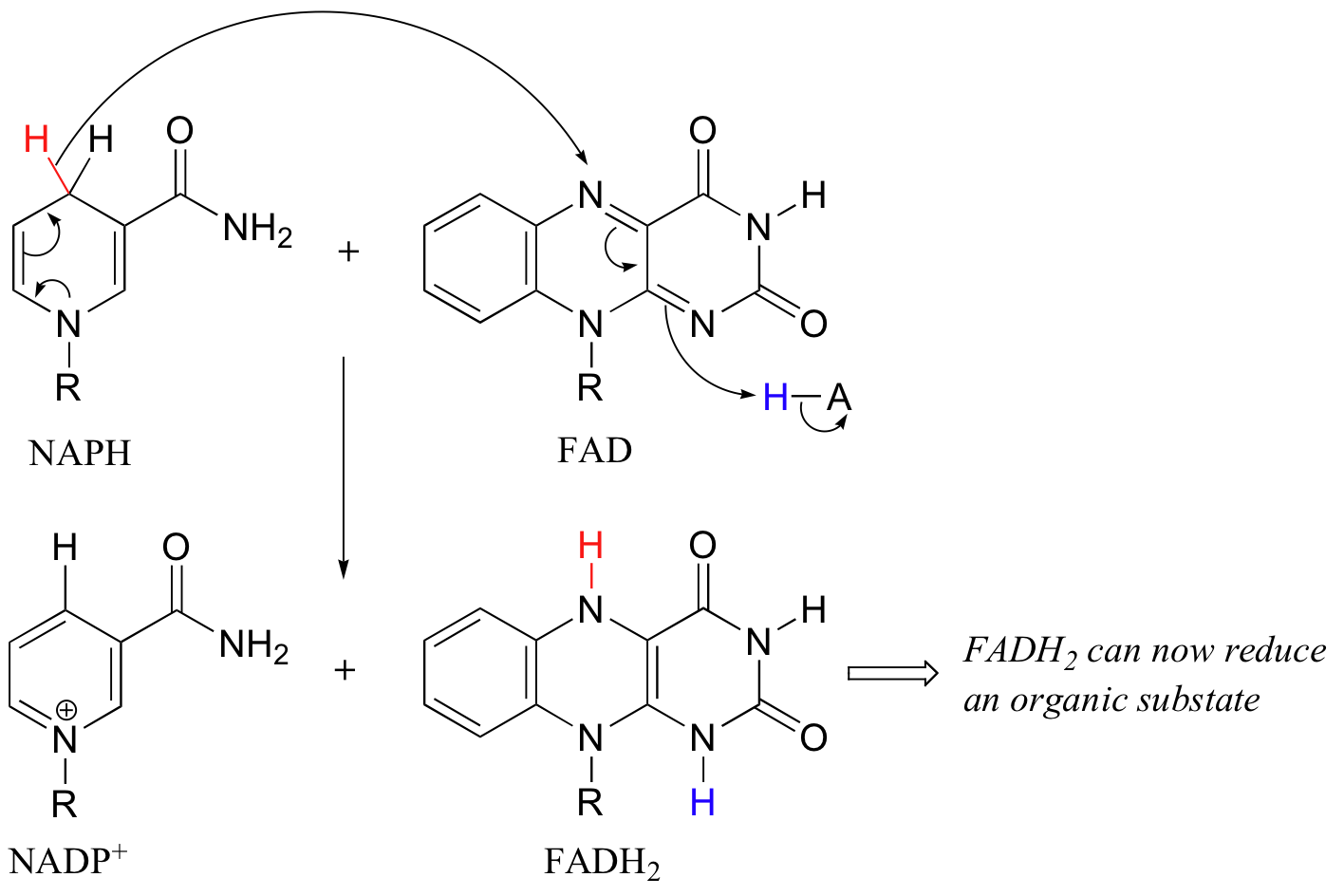
In this sense FADH2 is just a hydride carrier, obtaining its reducing hydride from NADPH and then transferring it on to an organic substrate. FAD then must be converted back to FADH2 before it can reduce another substrate molecule.
Until the 1990s, it was assumed that the oxidative pentose phosphate pathway was the source of NADPH for all species, but recent DNA sequencing projects have shown this to be incorrect. You have undoubtedly heard of the Human Genome Project, in which the entire DNA blueprint of human beings was sequenced, providing vast stores of information to biologists and medical researchers. What you may not be aware of is that the genomes of many other species have also been completely sequenced, including hundreds of microbial species. Among the microbial genomes that have been sequenced are those that belong to a classification of species called archaea which, although they resemble bacteria physically, are genetically a completely distinct kingdom of life. Interestingly, the genomes of archaea which have been sequenced so far do not seem to contain genes for the enzymes of the oxidative pentose phosphate pathway. These genes, and the enzymes they encode, are contained in the genomes of all other living things, as far as we know. Apparently, the archaeal microbes have some other means of regenerating NADPH from NADP+, most likely through one or more unique alternative glucose oxidation pathways.
The mountains of data provided by genome sequencing products are a treasure trove of information, not only for medical scientists trying to understand the genetic basis for human diseases, but also for biochemists interested in the diverse ways that enzymes have evolved to catalyze organic reactions.


