Making Diazonium Salts
- Page ID
- 3995
This page looks at the reaction between phenylamine (also known as aniline and aminobenzene) and nitrous acid - particularly its reaction at temperatures of less than 5°C to produce diazonium salts. If you want to know about the reactions of the diazonium ions formed, you will find a link at the bottom of the page.
The reactions of phenylamine with nitrous acid
Nitrous acid (also known as nitric(III) acid) has the formula HNO2. It is sometimes written as HONO to show the way it is joined up. Nitrous acid decomposes very readily and is always made in situ. In the case of its reaction with phenylamine, the phenylamine is first dissolved in hydrochloric acid, and then a solution of sodium or potassium nitrite is added. The reaction between the hydrochloric acid and the nitrite ions produces the nitrous acid.
\[ H^+ (aq) + NO_2^- (aq) \rightleftharpoons HNO_2 (aq)\]
Because nitrous acid is a weak acid, the position of equilibrium lies well the right. Phenylamine reacts with nitrous acid differently depending on the temperature.
The reaction on warming
If the mixture is warmed, you get a black oily product which contains phenol (amongst other things), and nitrogen gas is given off.
\[ CH_6H_5NH_2 + HNO_2 \rightarrow C_6H_5OH + H_2O + N_2\]
The reaction at low temperatures
The solution of phenylamine in hydrochloric acid (phenylammonium chloride solution) is stood in a beaker of ice. The sodium or potassium nitrite solution is also cooled in the ice. The solution of the nitrite is then added very slowly to the phenylammonium chloride solution - so that the temperature never goes above 5°C. You end up with a solution containing benzenediazonium chloride:
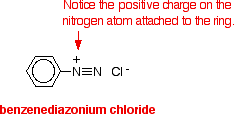
The positive ion, containing the -N2+ group, is known as a diazonium ion. The "azo" bit of the name refers to nitrogen. The ionic equation for the reaction is:
![]()
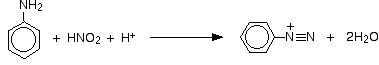
Notice that the chloride ions from the acid aren't involved in this in any way. If you use hydrochloric acid, the solution will contain benzenediazonium chloride. If you used a different acid, you would just get a different salt - a sulphate or hydrogensulphate, for example, if you used sulfuric acid.
The reactions of a diazonium salt are always done with a freshly prepared solution made in this way since the solutions do not keep. Diazonium salts are very unstable and tend to be explosive as solids.
Contributors
Jim Clark (Chemguide.co.uk)


