16.1: Common-Ion Effect in Acid–Base Equilibria
- Last updated
- Save as PDF
- Page ID
- 6464
↵
Learning Objectives
- Recognize common ions from various salts, acids, and bases.
- Calculate concentrations involving common ions.
- Calculate ion concentrations involving chemical equilibrium.
The common-ion effect is used to describe the effect on an equilibrium involving a substance that adds an ion that is a part of the equilibrium.
Introduction
The solubility products Ksp's are equilibrium constants in heterogeneous equilibria (i.e., between two different phases). If several salts are present in a system, they all ionize in the solution. If the salts contain a common cation or anion, these salts contribute to the concentration of the common ion. Contributions from all salts must be included in the calculation of concentration of the common ion. For example, a solution containing sodium chloride and potassium chloride will have the following relationship:
\[\mathrm{[Na^+] + [K^+] = [Cl^-]} \label{1} \]
Consideration of charge balance or mass balance or both leads to the same conclusion.
Common Ions
When \(\ce{NaCl}\) and \(\ce{KCl}\) are dissolved in the same solution, the \(\mathrm{ {\color{Green} Cl^-}}\) ions are common to both salts. In a system containing \(\ce{NaCl}\) and \(\ce{KCl}\), the \(\mathrm{ {\color{Green} Cl^-}}\) ions are common ions.
\[\mathrm{NaCl \rightleftharpoons Na^+ + {\color{Green} Cl^-}} \nonumber \]
\[\mathrm{KCl \rightleftharpoons K^+ + {\color{Green} Cl^-}} \nonumber \]
\[\mathrm{CaCl_2 \rightleftharpoons Ca^{2+} + {\color{Green} 2 Cl^-}} \nonumber \]
\[\mathrm{AlCl_3 \rightleftharpoons Al^{3+} + {\color{Green} 3 Cl^-}} \nonumber \]
\[\mathrm{AgCl \rightleftharpoons Ag^+ + {\color{Green} Cl^-}} \nonumber \]
For example, when \(\ce{AgCl}\) is dissolved into a solution already containing \(\ce{NaCl}\) (actually \(\ce{Na+}\) and \(\ce{Cl-}\) ions), the \(\ce{Cl-}\) ions come from the ionization of both \(\ce{AgCl}\) and \(\ce{NaCl}\). Thus, \(\ce{[Cl- ]}\) differs from \(\ce{[Ag+]}\). The following examples show how the concentration of the common ion is calculated.
Example \(\PageIndex{1}\)
What are \(\ce{[Na+]}\), \(\ce{[Cl- ]}\), \(\ce{[Ca^2+]}\), and \(\ce{[H+]}\) in a solution containing 0.10 M each of \(\ce{NaCl}\), \(\ce{CaCl2}\), and \(\ce{HCl}\)?
Solution
Due to the conservation of ions, we have
\[\mathrm{[Na^+] = [Ca^{2+}] = [H^+] = 0.10\: \ce M} \nonumber \]
but
\(\begin{alignat}{3}
\nonumber &\ce{[Cl- ]} &&= && && \:\textrm{0.10 (due to NaCl)}\\
\nonumber & && && + &&\mathrm{\:0.20\: (due\: to\: CaCl_2)}\\
\nonumber & && && + &&\mathrm{\:0.10\: (due\: to\: HCl)}\\
\nonumber & &&= && &&\mathrm{\:0.40\: M}
\nonumber \end{alignat}\)
Exercise \(\PageIndex{1}\)
John poured 10.0 mL of 0.10 M \(\ce{NaCl}\), 10.0 mL of 0.10 M \(\ce{KOH}\), and 5.0 mL of 0.20 M \(\ce{HCl}\) solutions together and then he made the total volume to be 100.0 mL. What is \(\ce{[Cl- ]}\) in the final solution?
Solution
\[\mathrm{[Cl^-] = \dfrac{0.1\: M\times 10\: mL+0.2\: M\times 5.0\: mL}{100.0\: mL} = 0.020\: M} \nonumber \]
Le Châtelier's Principle states that if an equilibrium becomes unbalanced, the reaction will shift to restore the balance. If a common ion is added to a weak acid or weak base equilibrium, then the equilibrium will shift towards the reactants, in this case the weak acid or base.
Example \(\PageIndex{2}\): Solubility of Lead Chloride
Consider the lead(II) ion concentration in this saturated solution of PbCl2. The balanced reaction is
\[ PbCl_{2 (s)} \rightleftharpoons Pb^{2+} _{(aq)} + 2Cl^-_{(aq)} \nonumber \]
Defining \(s\) as the concentration of dissolved lead(II) chloride, then:
\[[Pb^{2+}] = s \nonumber \]
\[[Cl^- ] = 2s \nonumber \]
These values can be substituted into the solubility product expression, which can be solved for \(s\):
\[\begin{eqnarray} K_{sp} &=& [Pb^{2+}] [Cl^-]^2 \\ &=& s \times (2s)^2 \\ 1.7 \times 10^{-5} &=& 4s^3 \\ s^3 &=& \frac{1.7 \times 10^{-5}}{4} \\ &=& 4.25 \times 10^{-6} \\ s &=& \sqrt[3]{4.25 \times 10^{-6}} \\ &=& 1.62 \times 10^{-2}\ mol\ dm^{-3} \end{eqnarray} \nonumber \]The concentration of lead(II) ions in the solution is 1.62 x 10-2 M. Consider what happens if sodium chloride is added to this saturated solution. Sodium chloride shares an ion with lead(II) chloride. The chloride ion is common to both of them; this is the origin of the term "common ion effect".
Look at the original equilibrium expression again:
\[ PbCl_2 \; (s) \rightleftharpoons Pb^{2+} \; (aq) + 2Cl^- \; (aq) \nonumber \]
What happens to that equilibrium if extra chloride ions are added? According to Le Châtelier, the position of equilibrium will shift to counter the change, in this case, by removing the chloride ions by making extra solid lead(II) chloride.
Of course, the concentration of lead(II) ions in the solution is so small that only a tiny proportion of the extra chloride ions can be converted into solid lead(II) chloride. The lead(II) chloride becomes even less soluble, and the concentration of lead(II) ions in the solution decreases. This type of response occurs with any sparingly soluble substance: it is less soluble in a solution which contains any ion which it has in common. This is the common ion effect.
Example \(\PageIndex{3}\)
If an attempt is made to dissolve some lead(II) chloride in some 0.100 M sodium chloride solution instead of in water, what is the equilibrium concentration of the lead(II) ions this time? As before, define s to be the concentration of the lead(II) ions.
\[[Pb^{2+}] = s \label{2} \]
The calculations are different from before. This time the concentration of the chloride ions is governed by the concentration of the sodium chloride solution. The number of ions coming from the lead(II) chloride is going to be tiny compared with the 0.100 M coming from the sodium chloride solution.
In calculations like this, it can be assumed that the concentration of the common ion is entirely due to the other solution. This simplifies the calculation.
So we assume:
\[[Cl^- ] = 0.100\; M \label{3} \]
The rest of the mathematics looks like this:
\begin{equation} \begin{split} K_{sp}& = [Pb^{2+}][Cl^-]^2 \\ & = s \times (0.100)^2 \\ 1.7 \times 10^{-5} & = s \times 0.00100 \end{split} \nonumber \end{equation}
therefore:
\begin{equation} \begin{split} s & = \dfrac{1.7 \times 10^{-5}}{0.0100} \\ & = 1.7 \times 10^{-3} \, \text{M} \end{split} \label{4} \end{equation}
Finally, compare that value with the simple saturated solution:
Original solution:
\[[Pb^{2+}] = 0.0162 \, M \label{5} \]
Solution in 0.100 M NaCl solution:
\[ [Pb^{2+}] = 0.0017 \, M \label{6} \]
The concentration of the lead(II) ions has decreased by a factor of about 10. If more concentrated solutions of sodium chloride are used, the solubility decreases further.
A Video Discussing Finding the Solubility of a Salt: Finding the Solubility of a Salt(opens in new window) [youtu.be]
Common Ion Effect with Weak Acids and Bases
Adding a common ion prevents the weak acid or weak base from ionizing as much as it would without the added common ion. The common ion effect suppresses the ionization of a weak acid by adding more of an ion that is a product of this equilibrium.
Adding a common ion to a system at equilibrium affects the equilibrium composition, but not the ionization constant.
The common ion effect of H3O+ on the ionization of acetic acid
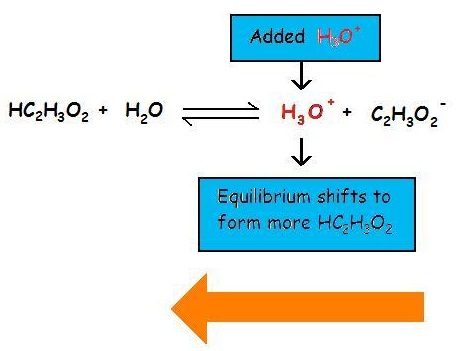
The common ion effect suppresses the ionization of a weak base by adding more of an ion that is a product of this equilibrium. Now consider the common ion effect of \(\ce{OH^{-}}\) on the ionization of ammonia
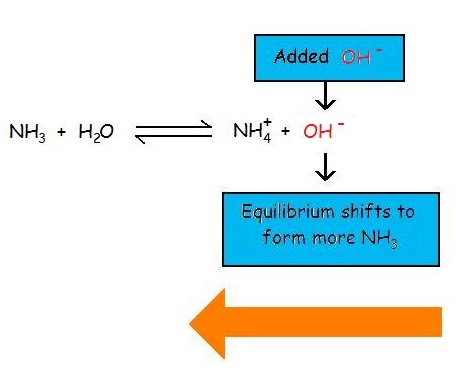
Adding the common ion of hydroxide shifts the reaction towards the left to decrease the stress (in accordance with Le Chatelier's Principle), forming more reactants. This decreases the reaction quotient, because the reaction is being pushed towards the left to reach equilibrium. The equilibrium constant, \(K_b=1.8 \times 10^{-5}\), does not change. The reaction is put out of balance, or equilibrium.
\[Q_a = \frac{\ce{[NH_4^{+}][OH^{-}]}}{\ce{[NH3]}} \nonumber \]
At first, when more hydroxide is added, the quotient is greater than the equilibrium constant. The reaction then shifts right, causing the denominator to increase, decreasing the reaction quotient and pulling towards equilibrium and causing Q to decrease towards K.
Common Ion Effect on Solubility
When a slightly soluble ionic compound is added to water, some of it dissolves to form a solution, establishing an equilibrium between the pure solid and a solution of its ions. For the dissolution of calcium phosphate, one of the two main components of kidney stones, the equilibrium can be written as follows, with the solid salt on the left:
\[\ce{Ca3(PO4)2(s) <=> 3Ca^{2+}(aq) + 2PO^{3−}4(aq)} \label{17.4.1} \]
As you will discover in more advanced chemistry courses, basic anions, such as S2−, PO43−, and CO32−, react with water to produce OH− and the corresponding protonated anion. Consequently, their calculated molarities, assuming no protonation in aqueous solution, are only approximate. The equilibrium constant for the dissolution of a sparingly soluble salt is the solubility product (Ksp) of the salt. Because the concentration of a pure solid such as Ca3(PO4)2 is a constant, it does not appear explicitly in the equilibrium constant expression. The equilibrium constant expression for the dissolution of calcium phosphate is therefore
\[K=\dfrac{[\mathrm{Ca^{2+}}]^3[\mathrm{PO_4^{3-}}]^2}{[\mathrm{Ca_3(PO_4)_2}]} \label{17.4.2a} \]
\[[\mathrm{Ca_3(PO_4)_2}]K=K_{\textrm{sp}}=[\mathrm{Ca^{2+}}]^3[\mathrm{PO_4^{3-}}]^2 \label{17.4.2b} \]
At 25°C and pH 7.00, \(K_{sp}\) for calcium phosphate is 2.07 × 10−33, indicating that the concentrations of Ca2+ and PO43− ions in solution that are in equilibrium with solid calcium phosphate are very low. The values of Ksp for some common salts vary dramatically for different compounds (Table E3). Although \(K_{sp}\) is not a function of pH in Equation \(\ref{17.4.2a}\), changes in pH can affect the solubility of a compound.
The solubility product expression tells us that the equilibrium concentrations of the cation and the anion are inversely related. That is, as the concentration of the anion increases, the maximum concentration of the cation needed for precipitation to occur decreases—and vice versa—so that \(K_{sp}\) is constant. Consequently, the solubility of an ionic compound depends on the concentrations of other salts that contain the same ions. This dependency is another example of the common ion effect where adding a common cation or anion shifts a solubility equilibrium in the direction predicted by Le Chatelier’s principle. As a result, the solubility of any sparingly soluble salt is almost always decreased by the presence of a soluble salt that contains a common ion.
Consider, for example, the effect of adding a soluble salt, such as CaCl2, to a saturated solution of calcium phosphate [Ca3(PO4)2]. We have seen that the solubility of Ca3(PO4)2 in water at 25°C is 1.14 × 10−7 M (Ksp = 2.07 × 10−33). Thus a saturated solution of Ca3(PO4)2 in water contains
- \(3 \times (1.14 \times 10^{−7}\; M) = 3.42 \times 10^{−7} M \; of \; Ca^{2+}\)
- \(2 \times (1.14 \times 10^{−7} M) = 2.28 \times 10^{−7} M \; of \; PO_4^{3−}\)
according to the stoichiometry shown in Equation \(\ref{17.4.2a}\) (neglecting hydrolysis to form HPO42−). If CaCl2 is added to a saturated solution of Ca3(PO4)2, the Ca2+ ion concentration will increase such that [Ca2+] > 3.42 × 10−7 M, making Q > Ksp. The only way the system can return to equilibrium is for the reaction in Equation \(\ref{17.4.2a}\) to proceed to the left, resulting in precipitation of Ca3(PO4)2. This will decrease the concentration of both Ca2+ and PO43− until Q = Ksp.
Note
Adding a common ion decreases solubility, as the reaction shifts toward the left to relieve the stress of the excess product. Adding a common ion to a dissociation reaction causes the equilibrium to shift left, toward the reactants, causing precipitation.
Example \(\PageIndex{5}\)
Consider the reaction:
\[ PbCl_2(s) \rightleftharpoons Pb^{2+}(aq) + 2Cl^-(aq) \nonumber \]
What happens to the solubility of PbCl2(s) when 0.1 M NaCl is added?
Solution
\[K_{sp}=1.7 \times 10^{-5} \nonumber \]
\[Q_{sp}= 1.8 \times 10^{-5}\nonumber \]
Identify the common ion: Cl-
Notice: Qsp > Ksp The addition of NaCl has caused the reaction to shift out of equilibrium because there are more dissociated ions. Typically, solving for the molarities requires the assumption that the solubility of PbCl2 is equivalent to the concentration of Pb2+ produced because they are in a 1:1 ratio.
Because Ksp for the reaction is 1.7×10-5, the overall reaction would be (s)(2s)2= 1.7×10-5. Solving the equation for s gives s= 1.62×10-2 M. The coefficient on Cl- is 2, so it is assumed that twice as much Cl- is produced as Pb2+, hence the '2s.' The solubility equilibrium constant can be used to solve for the molarities of the ions at equilibrium.
The molarity of Cl- added would be 0.1 M because Na+ and Cl- are in a 1:1 ration in the ionic salt, NaCl. Therefore, the overall molarity of Cl- would be 2s + 0.1, with 2s referring to the contribution of the chloride ion from the dissociation of lead chloride.
\[\begin{eqnarray} Q_{sp} &=& [Pb^{2+}][Cl^-]^2 \\ 1.8 \times 10^{-5} &=& (s)(2s + 0.1)^2 \\ s &=& [Pb^{2+}] \\ &=& 1.8 \times 10^{-3} M \\ 2s &=& [Cl^-] \\ &\approx & 0.1 M \end{eqnarray} \nonumber \]
Notice that the molarity of Pb2+ is lower when NaCl is added. The equilibrium constant remains the same because of the increased concentration of the chloride ion. To simplify the reaction, it can be assumed that [Cl-] is approximately 0.1M since the formation of the chloride ion from the dissociation of lead chloride is so small. The reaction quotient for PbCl2 is greater than the equilibrium constant because of the added Cl-. This therefore shift the reaction left towards equilibrium, causing precipitation and lowering the current solubility of the reaction. Overall, the solubility of the reaction decreases with the added sodium chloride.
Exercise \(\PageIndex{5}\)
Calculate the solubility of silver carbonate in a 0.25 M solution of sodium carbonate. The solubility of silver carbonate in pure water is 8.45 × 10−12 at 25°C.
Answer
2.9 × 10−6 M (versus 1.3 × 10−4 M in pure water)
A Video Discussing the Common Ion Effect in Solubility Products: The Common Ion Effect in Solubility Products (opens in new window) [youtu.be]
References
- Harwood, William S., F. G. Herring, Jeffry D. Madura, and Ralph H. Petrucci. General Chemistry Principles and Modern Applications. 9th ed. New Jersey: Prentice Hall, 2007.
Contributors and Attributions
- Emmellin Tung, Mahtab Danai (UCD)
- Jim Clark (ChemGuide)
Chung (Peter) Chieh (Professor Emeritus, Chemistry @ University of Waterloo)

